RESEARCH TEAM
The research will be carried out mainly at the University of Ioannina (UoI) by 2 Greek faculty members, 1 post-doc and 3 PhD students to be employed in the project. This core group is augmented by 3 scientists from abroad with expertise on the synthesis (M. Steinhart (UoO), H. Duran (MPI-P)) and surface characterization (H.-J. Butt (MPI-P)) of soft materials.
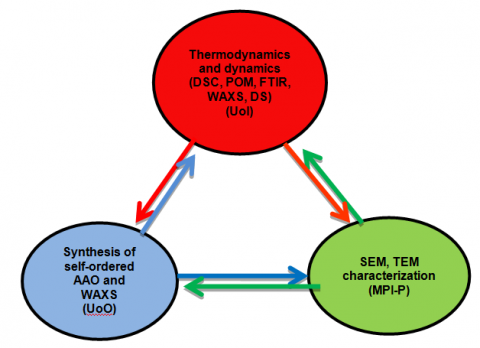
The RT from the University of Ioannina (UoI) (G. Floudas (PI), P. Papadopoulos, 1 post-doc, 3PhD students), with expertise in molecular and macromolecular self-assembly and dynamics, undertake WP1-5. Within these WPs they investigate the self-assembly by thermodynamic (differential scanning calorimetry) and structural means (X-rays, polarizing optical microscopy, FTIR) as well as the dynamics (with dielectric spectroscopy) in a range of systems that include crystallizable polymers, amphiphilic systems, liquid crystals and polypeptides under the hard confinement provided by nanoporous aluminum oxide templates. They investigate the effect of confinement/curvature on (i) the type of nucleation (homogeneous/heterogeneous) and overall crystallinity in crystallizable polymers, (ii) generating new nanostructures in amphiphilic molecules, (iii) the stability and coherence of smectic/nematic/discotic liquid crystalline phases and (iv) the self-organization and dynamics of polypeptides. There is a close collaboration with the UoO (M. Steinhart) with expertise on the electrochemical anodization of aluminum substrates as well as on self-organization under confinement and with the MPI-P (H. Duran, H.-J. Butt) with expertise on infiltration, surface modification and surface characterization of soft materials. The cooperativity of the research team is shown in Scheme I and the systems to be employed in Table 1.
Scheme I: Overview of the cooperative nature of the proposed work.
Table 1. Systems to be investigated within this project (the source of samples is also indicated).
1: Commercially available samples
2: Existing collaboration with Prof. N. Hadjichristidis and Prof. H. Iatrou (Univ. of Athens)[43][44]
3: Existing collaboration with Prof. K. Müllen (MPI-P) on bulk DLCs [40]and semifluorinated alkanes [34]
The research work packages and tasks, along with the corresponding deliverables, are described below.
WP1: Project coordination Duration: 36 m Researchers: G. Floudas (UoI) (more than 60% of the PIs research effort is devoted to this project)
Coordination of all WPs. Flow of systems and information. Evaluation of intermediate goals and possible re-adjustments. Planning of visits, organization of meetings, dissemination of results and procedures.
Deliverable: Coordination of research.
WP2: Crystallization under hard confinement Duration: 24 m
Researchers: PhD1 (UoI), Postdoc (UoI), M. Steinhart (UoO), H. Duran, H.-J. Butt (MPI-P), P. Papadopoulos (UoI), G. Floudas (UoI).
We investigate the type of nucleation (homogeneous/heterogeneous), crystal orientation and degree of crystallinity as a function of AAO pore size with DSC, POM, FTIR, X-rays as well as the local dynamics by dielectric spectroscopy (UoI). Complementary SEM is used for the surface characterization (MPI-P).
Deliverables: Type of nucleation, crystal orientation, degree of crystallinity and local dynamics under hard confinement. Design of polymeric nanofibers with tunable properties.
WP3: Engineering novel copolymer nanostructures under hard confinement Duration: 24 m
Researchers: PhD1 (UoI), Postdoc (UoI), M. Steinhart (UoO), H. Duran, H.-J. Butt (MPI-P), P. Papadopoulos (UoI), G. Floudas (UoI).
Task 3.1. The crystallizable/amorphous polyethylene oxide-b-polyisoprene system
Identify novel nanophases in comparison to the bulk for a range of PI-b-PEO copolymers32 as a function of the degree of segregation, composition and PEO molecular weight with DSC, X-rays, FTIR and TEM (MPI-P). Effect of confinement on PEO crystallization (in view of the results of WP2).
Deliverables: Identification of novel nanostructures, comparison to the bulk phase diagram.32
Task 3.2. Semi-fluorinated alkanes F(CF2)m(CH2)nH (FmHn)
We investigate the self-assembly and dynamics of model semifluorinated alkanes34 of the type FmHn under confinement as a function of the fluoroalkyl composition with DSC, POM, FTIR, X-rays and dielectric spectroscopy, respectively. We are particularly interested in exploring the effect of confinement/curvature on the stability of the intermediate phase (phase II) recently found in the symmetric F12H12 with the unexpected solid/liquid coexistence.34 We also investigate the dynamics of the uncompensated end C-F dipole pertinent to the local packing within the perfluoroalkyl environment.
Deliverables: Phase diagrams for several semi-fluorinated alkanes under confinement; stability of phase II; local dynamics within the CF2 phase.
WP4: Effect of confinement on the nematic/smectic/discotic phases of Liquid Crystals Duration: 36 m
Researchers: PhD2 (UoI), Postdoc (UoI), M. Steinhart (UoO), H. Duran, H.-J. Butt (MPI-P), P. Papadopoulos (UoI), G. Floudas (UoI).
Task 4.1 Calamitic Liquid Crystals
Detailed investigation of the effect of confinement on the type and coherence of the nematic-to-isotropic and smectic A-to-nematic phase transitions of the prototypical calamitic liquid crystals 5CB and the eutectic mixture E7. We employ DSC and FTIR, dielectric spectroscopy (DS) for the phase transitions under confinement and explore the critical nucleus size corresponding to the nematic and smectic A phases. X-ray scattering and DS are employed for the effect of surface-induced ordering (DS is a very sensitive probe of surface-induced molecular ordering). In addition, we investigate the effect of confinement on the molecular dynamics around the short and long molecules axes by probing the “slow” and “fast” dielectrically active processes.37 To this end, the high-frequency dielectric set-up will be employed. In a second step we investigate the effect of surface-induced ordering by modifying the surface of self-ordered AAO with phosphonate coupling molecules (surface functionalization at the MPI-P).
Deliverables: Stability of nematic and smectic A phases under confinement. Critical nucleus size for the nematic and smectic A phases. Effect of confinement on the glass-temperature. Effect of surface-induced ordering on the phase stability.
Task 4.2 Discotic Liquid Crystals (DLCs)
The phase state and disk dynamics of model DLCs of dipole-functionalized HBCs with record high charge carrier mobility - recently explored by members of the current RT40- are studied under hard confinement with DSC, X-rays, FTIR and DS in close collaboration with the synthesis group of Prof. K. Müllen. It was shown that these molecular systems possess a range of length- and time-scales (i.e. they exhibit polymer-like behavior) that will be affected by the confinement.
Deliverables: Phase state and charge carrier mobility of dipole-functionalized HBCs under hard confinement.
WP5: Effect of confinement on the type, correlation and dynamics of polypeptide secondary structures and copolypeptide nanostructures Duration: 36 m
Researchers: PhD3 (UoI), Postdoc (UoI), M. Steinhart (UoO), H. Duran, H.-J. Butt (MPI-P), P. Papadopoulos (UoI), G. Floudas (UoI).
Task 5.1 Homopolypeptides bearing different secondary structures
We investigate the effect of confinement/curvature on the type and coherence of different peptide secondary structures with X-rays, FTIR, DS and DSC. In-situ polymerization within the self-ordered AAO is made by H. Duran (MPI-P). Wide-angle X-ray indentifies both the type and local packing of secondary structures, whereas DS provides the persistence of α-helical motifs. The systems include polypeptides bearing a single secondary structure (i.e., β-sheets of polyglycine (PGly), α-helices of poly(ε-carbobenzyloxy-L-lysine) (PZLL)) and polypeptides bearing both (polyalanine (Pala), low molecular weight PBLG) secondary structures.
Deliverables: Stability-control of the type, orientation and persistence of peptide secondary structures.
Task 5.2 Copolypeptides bearing different nanostructures and peptide secondary structures
Here we investigate (with X-rays, DS and DSC) the effect of confinement on block copolypeptides based on PBLG-b-PGly and PBLG-b-Pala, i.e. blocks with different peptide secondary structures. In these systems there is a competition for ordering over multiple length scales (peptide secondary structure, nanophase separation, AAO walls). The synthesis of such model systems has already been reported (by H. Iatrou, N. Hadjichristidis at the Univ. of Athens (UoA)) and the bulk phase state and dynamics have been examined by members of the current RT in collaboration with the UoA.43,44
Deliverables: Copolypeptide phase state and peptide secondary structures under hard confinement.
Conclusions .
SOFT-CONFINE is positioned at the interface of well-established hitherto not cross-fertilizing fields with strong impact to nanocomposites, display industry, microelectronics and the biological world. It involves novel fabrication and characterization strategies that will enable us to achieve a fundamental understanding of the effect of confinement in controlling nucleation, crystal orientation and growth, the associated phase and phase transitions, and the dynamics of model soft materials that include crystallizable polymers, amphiphilic molecules, liquid crystals and biopolymers. This unified research approach will open up new routes for both scientific and technological research by tackling simultaneously structural, thermodynamic and dynamic issues over a broad spatio-temporal range. The research program will be implemented at the UoI by an interdisciplinary team headed by the PI, 1 staff scientist, 3 PhD students, 1 post-doc and 3 leading scientists from abroad with expertise in the fields of nanoporous materials and surface characterization. We envisage significantly contributing to the new emerging field of soft matter under hard confinement, where the many outstanding challenges are only now just being addressed.
References
[1] Shin, K.; Xiang, H.; Moon, S.I.; Kim, T.; McCarthy, T.J.; Russell, T.P. Science 2004, 306, 76.
[2] Stainhart, M. Adv. Polym. Sci. 2008, 220, 123.
[3] Martin, C.R. Science 1994, 266, 1961.
[4] Cai, Z.; Martin, C. R. J. Am. Chem. Soc. 1989, 111, 4138.
[5] Liang, W.; C. R. Martin, C. R. J. Am. Chem. Soc. 1990, 112, 9666.
[6] Hou, S. F.; Wang, J. H.; Martin, C. R. Nano Lett. 2005, 5, 231.
[7] Lakshmi, B. B.; Martin, C. R. Nature 1997, 388, 758.
[8] Lee, S. B.; Mitchell, D. T.; Trofin, L. et al. Science 2002, 296, 2198.
[9] Dai, J. H.; Baker, G. L.; Bruening, M. L. Anal. Chem. 2006, 78, 135.
[10] Kohli, P.; Harrell, C. C.; Cao, Z. H.; Gasparac, R., Tan, W. H.; Martin, C. R. Science 2004, 305, 984.
[11] Yu, B.; Sun, P.; Chen, T.; Jin, Q.; Ding, D.; Li, B.; Shi, A.-C. Phys. Rev. Lett. 2006, 96, 138306.
[12] Yu, B.; Sun, P.; Chen, T.; Jin, Q.; Ding, D.; Li, B.; Shi, A.-C. J. Chem. Phys. 2007, 127, 114906.
[13] Crawford, G.P.; Steele, L.M.; Ondriscrawford, R. et al. J. Chem. Phys. 1992, 96, 7788.
[14] Masuda, H.; Fukuda, K. Science 1995, 268, 1466.
[15] Schmelzer, J.W.P.; Abyzov, A.S. J. Chem. Phys. 2011, 134, 054511-1.
[16] Wang, H.; Keum, J.K.; Heltner, A. et al. Science 2009, 323, 757.
[17] Ma, Y.; Hu, W.; Hobbs, J.; Reiter, G. Soft Matter 2008, 4, 540-543.
[18] Steinhart, M.; Göring, P.; Dernaika, H. et al. Phys. Rev. Lett. 2006, 97, 027801-1.
[19] Massa, M.V.; Dalnoki-Veress, K. Phys. Rev. Lett. 2004, 92, 255509-1.
[20] Röttele, A.; Thurn-Albrecht, T.; Sommer, J.-U.; Reiter, G. Macromolecules 2003, 36, 1257-1260.
[21] Loo, Y.-L.; Register, R.A.; Ryan, A.J. Phys. Rev. Lett. 2000, 84, 4120-4123.
[22] Woo, E.; Huh, J.; Jeong, Y.G.; Shin, K. Phys. Rev. Lett. 2007, 98, 136103-1.
[23] Duran, H.; Stainhart, M.; H.-J. Butt, Floudas, G. Nano Lett. 2011, 11, 1671.
[24] Jin, Y.; Rogunova, M.; Hiltner, A. et al. Polym. Sci., Polym. Phys. Ed. 2004, 42, 3380-3396.
[25] Block Copolymers, Hadjichristidis, N.; Pispas, S.; Floudas, G. J. Wiley and Sons Inc. 2002.
[26] Li, W.; Wickham, R.A. Macromolecules 2006, 39, 8492.
[27] Li, W.; Wickham, R.A.; Garbary, R.A. Macromolecules 2006, 39, 806.
[28] Erukhimovich, I.; Johner, A. EPL 2007, 79, 56004.
[29] Xiang, H.; Shin, K.; Kim, T.; Moon, S.I.; McCarthy, T.J.; Russell, T.P. Macromolecules 2004, 37, 5660.
[30] Xiang, H.; Shin, K.; Kim, T.; Moon, S.I. et al. J. Polym. Sci., Polym. Phys. 2005, 43, 3377.
[31] Sun, Y.M.; Steinhart, M.; Zschech, D. et al. Macromol. Rapid. Commun. 2005, 26, 369.
[32] Floudas, G.; Vazaiou, B.; Schipper, F.; Ulrich, R.; Wiesner, U. et al. Macromolecules 2001, 34, 2947.
[33] Russell, T.P.; Rabolt, J.F. Macromolecules 1986, 19, 1135.
[34] Nunez, E.; Clark, C.G.; Cheng, W.; Best, A. Floudas, G. et al. J. Phys. Chem. B 2008, 112, 6542.
[35] Wilms, D.; Winkler, A.; Virnau, P.; Binder, K. Phys. Rev. Lett. 2010, 105, 4.
[36] Schilling, T.; Frenkel, D., Phys. Rev. Lett. 2004, 92,8.
[37] Rozanski, S. A.; Stannarius, R.; Groothues, H.; Kremer, F. Liq. Cryst. 1996, 20, 59.
[38] Iannacchione, G. S.; Finotello, D., Phys. Rev. Lett. 1992, 69, 2094.
[39] Grigoriadis, C.; Duran, H.; Steinhart, M.; Butt, H.-J.; Floudas, G. in preparation.
[40] Haase, N.; Grigoriadis, C.; Butt, H.-J.; Müllen, K.; Floudas, G. J. Phys. Chem. B 2011, 115, 5807.
[41] Floudas, G.; Spiess, H.W. Macromol. Rapid. Commun. 2009, 30, 278.
[42] Duran, H.; Gitsas, A.; Floudas, G. et al. Macromolecules 2009, 42, 2881.
[43] Papadopoulos, P.; Floudas, G.; Schnell, I. et al. Biomacromolecules 2005, 6, 2352.
[44] Gitsas, A.; Floudas, G.; Mondeshki, M.; Spiess, H.W. Macromolecules 2008, 41, 8072.